Understanding the Difference Between Buprenorphine and Suboxone
Is buprenorphine the same as Suboxone? No, they are not the same—but they are closely related.
Buprenorphine is the active medication ingredient that treats opioid use disorder. Suboxone is a brand-name product that combines buprenorphine with naloxone, an ingredient designed to deter misuse.
Here’s the quick breakdown:
- Buprenorphine = Single medication (partial opioid agonist)
- Suboxone = Buprenorphine + Naloxone (combination product)
- Subutex = Brand-name buprenorphine-only product (discontinued in 2011, generics available)
Think of it this way: buprenorphine is the core treatment ingredient, while Suboxone is one specific formulation containing it. All Suboxone contains buprenorphine, but not all buprenorphine products are Suboxone.
If you’re researching treatment options for opioid use disorder, understanding this distinction is vital. This confusion is common, especially when making informed decisions about your care. With overdose deaths increasing annually since 1999, choosing the right medication-assisted treatment is crucial.
I’m Dr. Chad Elkin, a board-certified addiction medicine physician and founder of National Addiction Specialists. A frequent question I hear is “is buprenorphine the same as Suboxone?” Clearing up this confusion helps patients make better treatment decisions. In this guide, I’ll break down how these medications compare and help you understand which option might work best for you.
Is buprenorphine the same as suboxone word roundup:
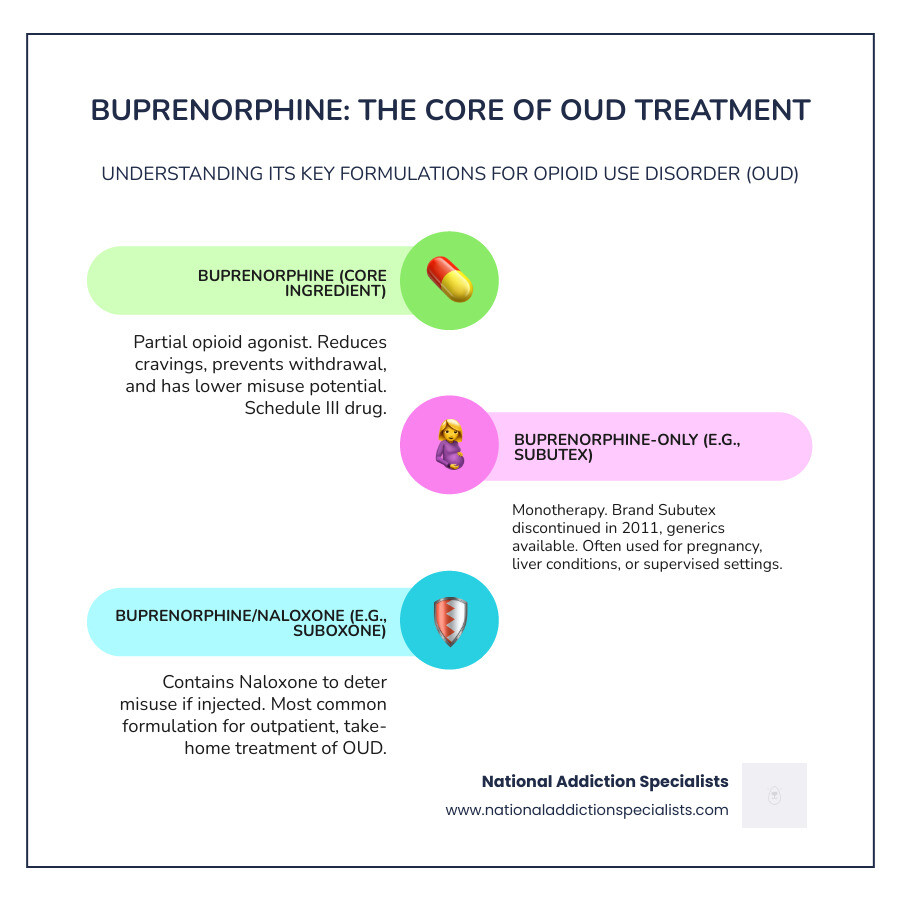
What is Buprenorphine? The Core Ingredient
When you’re researching is buprenorphine the same as Suboxone, you first need to understand what buprenorphine actually is. Think of it as the foundation—the core ingredient that makes treatment for opioid use disorder possible.
Buprenorphine is a prescription medication classified as a partial opioid agonist. Now, that’s medical jargon, but here’s what it means in plain English: buprenorphine works on the same receptors in your brain as other opioids, but it doesn’t fully activate them the way heroin or oxycodone would.

Imagine your brain’s opioid receptors as dimmer switches. Full opioids turn the lights all the way up—creating intense euphoria but also dangerous side effects and high overdose risk. Buprenorphine, on the other hand, turns the lights up just enough to reduce withdrawal symptoms and cravings, without flooding your system with that intense high.
This is where buprenorphine’s ceiling effect becomes incredibly important. After a certain dose, taking more buprenorphine doesn’t increase its opioid effects. This built-in safety feature significantly lowers the potential for misuse and respiratory depression—the dangerous slowing of breathing that causes most opioid overdose deaths.
Because of how buprenorphine works, it accomplishes two critical goals: it reduces cravings for opioids and prevents withdrawal symptoms. This allows people in recovery to stabilize their lives without the constant physical need driving them back to dangerous opioid use. The medication essentially quiets the storm in your brain, giving you space to focus on healing and rebuilding.
Due to its effectiveness and lower misuse potential compared to full opioid agonists, buprenorphine is classified as a Schedule III controlled substance. This means it’s recognized as having legitimate medical use with a moderate to low potential for physical and psychological dependence.
You can learn more about how buprenorphine works in our comprehensive guide: More info about Buprenorphine. The Substance Abuse and Mental Health Services Administration (SAMHSA) also provides detailed information: What is Buprenorphine?.
Buprenorphine-Only Medications (Subutex and Generics)
While Suboxone combines buprenorphine with naloxone, buprenorphine can also be prescribed by itself. This is called buprenorphine monotherapy—meaning it’s the only active ingredient.
You might have heard of Subutex, which was the original brand-name version of buprenorphine-only medication. However, the brand-name Subutex was discontinued in the United States in 2011. The good news? Generic versions of buprenorphine monotherapy are still available today.
So when would a doctor prescribe buprenorphine without naloxone? There are specific situations where buprenorphine-only products make more sense than combination medications.
Pregnancy is the most common reason for prescribing buprenorphine monotherapy. Treating opioid use disorder during pregnancy is absolutely critical—it protects both the mother and the developing baby from the dangers of continued opioid use. While naloxone is generally considered safe during pregnancy, many providers prefer the simplicity of buprenorphine alone to eliminate any theoretical concerns.
Some individuals may have sensitivity to naloxone or experience adverse reactions to it. Though this is rare, these patients need a buprenorphine-only option to continue their treatment safely.
In supervised settings—like during the initial induction phase when you’re first starting treatment, or in inpatient facilities—healthcare providers might use buprenorphine-only products. Since trained medical staff are watching the situation and the risk of misuse is minimal, the abuse-deterrent properties of naloxone become less necessary.
Patients with certain liver conditions may also be better candidates for buprenorphine monotherapy, as their healthcare provider can more precisely adjust dosing without the additional component.
Understanding these differences helps clarify why is buprenorphine the same as Suboxone isn’t just a simple yes or no question—the answer depends on which specific formulation is being used and why. For more details on these distinctions, visit our page: More on Suboxone vs Subutex.
Is Buprenorphine the Same as Suboxone? A Detailed Comparison
Is buprenorphine the same as Suboxone? The short answer is no—but they’re intimately connected. Think of buprenorphine as the essential ingredient and Suboxone as a recipe that adds an important safety feature.
The opioid epidemic continues to devastate communities. From 1999 to 2021, opioid overdose deaths increased every year. In 2019 alone, about 600,000 people died from drug use, with 80% of these deaths related to opioids. These aren’t just statistics—they represent people and families searching for hope.
That’s exactly why medications like Suboxone were developed. Suboxone represents what we call combination therapy—it pairs buprenorphine with naloxone to create an abuse-deterrent formulation. In practical terms, while buprenorphine treats your opioid use disorder, naloxone acts as a built-in safety mechanism, which is especially important for patients managing their medication at home.
Naloxone is an opioid antagonist, meaning it blocks opioid effects. When you take Suboxone as prescribed—dissolving it under your tongue or against your cheek—the naloxone is barely absorbed and remains inactive. But if someone tries to misuse Suboxone by injecting or snorting it, the naloxone springs into action. It blocks the opioid receptors, triggering precipitated withdrawal—an immediate, intense, and unpleasant withdrawal. This design makes Suboxone significantly safer for take-home treatment compared to buprenorphine-only products.
Here are the key differences:
| Feature | Buprenorphine-Only Products | Buprenorphine/Naloxone Products |
|---|---|---|
| Active Ingredients | Buprenorphine only | Buprenorphine + Naloxone |
| Primary Use | OUD treatment in supervised settings, pregnancy, pain management (some formulations) | OUD treatment, especially outpatient maintenance |
| Abuse Deterrence | Lower inherent protection against misuse | Strong deterrent due to naloxone component |
| Common Forms | Sublingual tablets, monthly injections (Sublocade), implants (Probuphine) | Sublingual films (most common), sublingual tablets, buccal films |
| Typical Use Setting | Initial induction phase, highly supervised environments, pregnancy cases | Outpatient maintenance, take-home prescriptions |
Active Ingredients: Is buprenorphine the same as Suboxone?
No, because of what’s inside each medication.
Buprenorphine is the primary active drug doing the work of treating opioid use disorder. As a partial opioid agonist, it occupies your opioid receptors to reduce withdrawal and cravings, while blocking other opioids’ effects. It is the foundation of the treatment.
Naloxone is the secondary ingredient found only in Suboxone and similar combination products. As mentioned, its sole purpose is to discourage misuse. When taken correctly, it remains inactive. If the medication is injected or snorted, the naloxone activates and causes precipitated withdrawal, making misuse unpleasant.
This thoughtful design provides built-in protection for patients managing their recovery at home. If you want to dive deeper into how this combination medication works, we’ve created a comprehensive resource: What is Suboxone?.
Administration and Forms
The way you take your medication matters—a lot. Both buprenorphine-only and combination products come in several forms, each designed with specific treatment goals in mind.

Sublingual films are probably what most people picture when they think of Suboxone. These thin strips dissolve under your tongue, allowing the medication to absorb directly into your bloodstream. This route bypasses your digestive system, which is important because swallowing naloxone would render it largely ineffective. Generic versions of buprenorphine/naloxone also come in this convenient film form.
Sublingual tablets work the same way—they dissolve under your tongue for direct absorption. While brand-name Suboxone tablets have been discontinued, generic buprenorphine/naloxone tablets are still widely available. Buprenorphine-only tablets can also be prescribed in certain situations.
Buccal films offer another option, placed between your cheek and gum to dissolve. The absorption method is similar to sublingual, just in a different location in your mouth.
For patients who prefer not to take daily medication or struggle with adherence, monthly injections like Sublocade provide an excellent alternative. These buprenorphine-only extended-release injections are administered by a healthcare professional once a month, eliminating the need for daily dosing and reducing the risk of diversion or misuse.
Six-month implants (Probuphine) were another long-acting option that provided steady medication delivery for half a year. While they required a minor surgical procedure for insertion and removal, they offered significant convenience for appropriate candidates.
One important thing you should know: any buprenorphine medication that dissolves in your mouth—whether film or tablet—has been associated with dental problems like tooth decay, cavities, and tooth loss. The FDA has issued warnings about this risk. Good oral hygiene is essential if you’re taking these medications. Rinse your mouth with water after the film or tablet dissolves, wait at least an hour before brushing, and see your dentist regularly. You can read more about this important safety information here: FDA warning on dental problems with buprenorphine.
Side Effects and Risks
Let’s talk honestly about what you might experience while taking these medications. Because buprenorphine is the primary active ingredient in both buprenorphine-only products and Suboxone, their side effect profiles are very similar.
Common side effects you might notice include constipation (this is probably the most frequent complaint), headaches, nausea and vomiting, drowsiness or sedation, dizziness, sweating (sometimes excessive), dry mouth, body aches, difficulty sleeping or unusual dreams, and mood changes. Most of these are mild and often improve as your body adjusts to the medication over the first few weeks. Don’t suffer in silence—let your healthcare provider know if any side effect becomes bothersome or doesn’t go away.
Now for the more serious risks you need to know about. Respiratory depression—slowed or difficult breathing—can occur with buprenorphine, though it’s far less likely than with full opioid agonists thanks to that ceiling effect we discussed earlier. The real danger comes when buprenorphine is combined with other central nervous system (CNS) depressants like alcohol, benzodiazepines (medications like Xanax, Valium, or Klonopin), or certain sedatives. This combination dramatically increases the risk of severe respiratory depression, coma, or even death. I cannot stress this enough: never combine these substances unless you’re under close medical supervision and your doctor has specifically approved it.
Liver problems can sometimes develop during buprenorphine treatment. That’s why we monitor liver function with blood tests before starting treatment and periodically throughout. Watch for warning signs like yellowing of your skin or eyes, dark-colored urine, or severe abdominal pain, and report them immediately.
If you’re pregnant or planning to become pregnant, we need to discuss Neonatal Opioid Withdrawal Syndrome (NOWS). Babies born to mothers taking buprenorphine or Suboxone may experience withdrawal symptoms after birth. However—and this is crucial—treating your opioid use disorder during pregnancy with buprenorphine is generally much safer for both you and your baby than untreated opioid use disorder. We’ll monitor you carefully and ensure your baby gets appropriate care after delivery.
Other serious but less common risks include adrenal insufficiency (where your adrenal glands don’t produce enough hormones) with long-term use, and severe allergic reactions (though these are rare). If you develop a rash, itching, swelling, severe dizziness, or trouble breathing, seek immediate medical attention.
Your safety is our top priority. That’s why we take a complete medical history and review all your current medications—prescription, over-the-counter, and supplements—before starting treatment. Open communication with your healthcare team is essential for safe, effective treatment.
How Buprenorphine and Suboxone Fit into Medication-Assisted Treatment (MAT)
Understanding is buprenorphine the same as Suboxone becomes even more important when you see how both medications work within the broader framework of treatment. Neither buprenorphine nor Suboxone exists in isolation—they’re powerful tools within what we call Medication-Assisted Treatment, or MAT.
MAT is an evidence-based approach that treats the whole person, not just the addiction. Think of it as a three-legged stool: medication, counseling, and behavioral support. Remove any one leg, and the stool becomes unstable. The medication manages the physical aspects of opioid dependence, while counseling and behavioral therapies address the psychological, emotional, and social factors that contribute to substance use.
Here’s what makes MAT so effective: the medication reduces withdrawal symptoms so you’re not constantly battling physical discomfort. It decreases cravings, those intense urges that can derail even the strongest commitment to recovery. And because buprenorphine occupies your opioid receptors, it blocks the euphoric effects of other opioids—meaning if someone were to relapse, they wouldn’t get the “high” they’re seeking. This removes much of the reward that drives continued use.
With these physical barriers managed, you’re free to focus on the real work of recovery. You can participate fully in counseling sessions, engage with support groups, rebuild relationships, and develop healthy coping strategies. This is where lasting change happens.
The research backs this up overwhelmingly. MAT significantly reduces illicit opioid use, improves patient survival rates, and increases the likelihood of sustained recovery. Studies show lower all-cause and overdose mortality among people receiving MAT, along with improved quality of life and social functioning. For more insight into how this comprehensive approach works in practice, visit How Suboxone Treatment Works and Benefits of Suboxone Treatment.
Starting Treatment: The Induction Phase
Starting buprenorphine or Suboxone requires careful timing and medical supervision, especially during what we call the “induction phase.” This first step is crucial, and getting it right makes all the difference in your comfort and success.
The main concern during induction is avoiding something called “precipitated withdrawal”—and trust me, you want to avoid this. Here’s what happens: buprenorphine has a very strong attraction to your opioid receptors. When you take it, it essentially pushes any other opioids off those receptors and takes their place. If there’s still a significant amount of full opioid in your system when you take your first buprenorphine dose, this sudden displacement can trigger rapid, intense withdrawal symptoms that are far worse than natural withdrawal.
To prevent this uncomfortable experience, we need you to already be in mild to moderate withdrawal before taking your first dose. I know that sounds counterintuitive—why would we want you to feel worse before you feel better? But this waiting period is essential for your safety and comfort.
The timing depends on what type of opioid you’ve been using. For short-acting opioids like heroin, oxycodone, or fentanyl, you typically need to wait at least 12 hours since your last use. For long-acting opioids like methadone or extended-release formulations, the waiting period is much longer—often 24 hours for most long-acting prescription opioids, and sometimes up to 72 hours for methadone.
During this phase, our healthcare providers will carefully assess your withdrawal symptoms using standardized scales. We start with a low dose and gradually increase it until your symptoms are well-controlled. This measured approach helps us find the right dosage for your specific needs while minimizing any discomfort.
You won’t go through this alone. Medical supervision during induction ensures you’re safe, comfortable, and starting your treatment journey on the right foot. Learn more about what to expect during this process at How Does Suboxone Treatment Work?.
Frequently Asked Questions about Buprenorphine and Suboxone
We know you might have more questions, especially given the nuances of these medications. Here, we address some of the most common inquiries we receive.
Why was the brand-name Subutex discontinued in the U.S.?
The brand-name Subutex, which contained only buprenorphine, was discontinued in the U.S. in 2011. This decision was largely driven by a shift in medical practice towards abuse-deterrent formulations. Suboxone, with its combination of buprenorphine and naloxone, became the preferred alternative for most outpatient settings. The naloxone component in Suboxone acts as a deterrent to misuse by causing precipitated withdrawal if injected, making it a safer option for take-home doses.
While the brand-name Subutex is no longer available, generic buprenorphine-only tablets are still manufactured and prescribed in specific situations, such as for pregnant patients or individuals with a documented sensitivity to naloxone, as discussed earlier.
Can you misuse buprenorphine or Suboxone?
Yes, like any opioid medication, buprenorphine and Suboxone can be misused, though their abuse potential is significantly lower than that of full opioid agonists.
- Buprenorphine-only products: These can be misused, particularly if injected or snorted, to achieve a partial opioid effect. However, buprenorphine’s “ceiling effect” means that even with misuse, the euphoric effects are limited, and the risk of severe respiratory depression is lower than with full opioids.
- Suboxone (buprenorphine/naloxone): The addition of naloxone is a key abuse deterrent. If Suboxone is injected or snorted, the naloxone component becomes active and can trigger immediate, unpleasant precipitated withdrawal symptoms. This mechanism is specifically designed to discourage misuse by injection. When taken as prescribed (sublingually or buccally), the naloxone is inactive, and the buprenorphine works effectively without causing withdrawal.
While misuse is possible, the overall risk of overdose is much lower with buprenorphine products compared to full opioid agonists, especially when used under medical supervision as part of a comprehensive MAT program.
What are the key differences when asking “is buprenorphine the same as Suboxone”?
The main difference between buprenorphine and Suboxone boils down to the presence of naloxone and their resulting formulations and typical use cases.
- Naloxone: Suboxone contains naloxone; buprenorphine-only products do not. This is the single most important distinction.
- Combination vs. Single Ingredient: Suboxone is a combination product (buprenorphine + naloxone), designed with an abuse-deterrent feature. Buprenorphine is the single-ingredient medication.
- Brand Names and Formulations: While “buprenorphine” refers to the active drug, it comes in various brand-name and generic forms. Suboxone is a specific brand name for a buprenorphine/naloxone combination, typically a sublingual film. Other buprenorphine/naloxone brands include Zubsolv (sublingual tablets) and Bunavail (buccal film). Buprenorphine-only products include generic sublingual tablets, as well as extended-release injections like Sublocade and implants like Probuphine (now discontinued).
- Typical Use Setting: Suboxone is widely used for maintenance treatment in outpatient settings due to its abuse-deterrent properties. Buprenorphine-only products are often reserved for specific populations (e.g., pregnant individuals) or highly supervised settings.
Take Control of Your Recovery Journey
If you’ve made it this far, you’ve taken an important step—understanding your treatment options. Now you know that buprenorphine is the active ingredient that does the heavy lifting in treating opioid use disorder, while Suboxone is a thoughtfully designed formulation that combines buprenorphine with naloxone for added safety and misuse deterrence.
But knowledge alone isn’t recovery. The real change happens when you take action.
Your recovery journey is deeply personal. What works for one person might not be the perfect fit for another. That’s why personalized treatment is so important. Some people do best with buprenorphine-only products, while others benefit from the combination approach of Suboxone. The right choice depends on your unique situation, medical history, and treatment goals.
At National Addiction Specialists, we understand that seeking help takes courage. We’ve built our practice around making that step as comfortable and accessible as possible. Through our telemedicine-based care, you can receive expert Suboxone treatment from the privacy and convenience of your own home in Tennessee or Virginia. No waiting rooms, no judgment—just confidential, professional care when and where you need it.
Our team of addiction medicine specialists works with you to create a treatment plan custom to your life. We combine medication-assisted treatment with counseling and support, addressing not just the physical aspects of opioid dependence, but the whole picture of your recovery. Whether you’re just starting to explore treatment options or you’re ready to begin today, we’re here to guide you.
We accept Medicaid and Medicare, because everyone deserves access to quality addiction treatment regardless of their financial situation. For a deeper understanding of all your treatment options and what to expect, we invite you to read: The Complete Guide to Suboxone Treatment Options.
Recovery is possible. You don’t have to figure this out alone.
Make an Appointment to Treat Addiction
Please don’t hesitate. Make an appointment today.
This article was medically reviewed by:
Chad Elkin, MD, DFASAM is a board-certified addiction medicine physician, founder, and Chief Medical Officer of National Addiction Specialists, dedicated to treating substance use disorders. A Distinguished Fellow of the American Society of Addiction Medicine (ASAM), Dr Elkin currently serves as President of the Tennessee Society of Addiction Medicine (TNSAM) and has held various leadership roles within the organization. Dr Elkin chairs ASAM’s Health Technology Subcommittee and is an active member of its Practice Management and Regulatory Affairs Committee, State Advocacy and Legislative Affairs Committee, and other committees. He also serves on the planning committee for the Vanderbilt Mid-South Addiction Conference. Committed to advancing evidence-based policy, Dr Elkin is Chairman of the Tennessee Association of Alcohol, Drug, & Other Addiction Services (TAADAS) Addiction Medicine Council, which collaborates with the TN Department of Mental Health & Substance Abuse Services (TDMHSAS). He has contributed to numerous local, state, and national task forces, helping develop professional guidelines, policies, and laws that align with best practices in addiction medicine. His work focuses on reducing addiction-related harm, combating stigma, and ensuring access to effective treatment. Passionate about the field of addiction medicine, he remains dedicated to shaping policy and enhancing patient care.
Suboxone® and Subutex® are a registered trademark of Indivior UK Limited. Any mention and reference of Suboxone® and Subutex® in this website is for informational purposes only and is not an endorsement or sponsorship by Indivior UK Limited.








