How to Get Your Suboxone Prior Authorization Approved Fast
Why Suboxone Prior Authorization Matters for Your Treatment
Suboxone prior authorization is a health plan requirement where your insurance reviews and approves your Suboxone prescription before covering it. This process ensures the medication is medically necessary and meets specific criteria set by your insurance company.
Quick Answer: What You Need to Know
| Aspect | Key Information |
|---|---|
| What It Is | Insurance approval required before Suboxone coverage begins |
| Typical Timeline | 24 hours to 3 business days for most plans |
| Main Requirements | Opioid use disorder diagnosis, qualified prescriber with DEA X-prefix, counseling enrollment, urine drug screens |
| Common Dosage Limits | Up to 16-24mg/day initially; higher doses require additional justification |
| Authorization Periods | 30 days to 12 months depending on insurance and treatment phase |
Why This Process Exists
Research shows that 32 of 50 states require prior authorization for at least one buprenorphine formulation. While these requirements aim to ensure appropriate use and prevent misuse, they also create barriers to life-saving treatment. The American Medical Association strongly urges states to remove these barriers because delays in accessing Suboxone can increase overdose risk and discourage people from seeking help.
Understanding the Impact
Prior authorization affects vulnerable populations most. Studies found that 15 states require authorization even for buprenorphine-naloxone (Suboxone), a first-line treatment. These requirements often include restrictive surveillance measures like mandatory urine screenings, dosage maximums that interfere with medical decisions, and counseling requirements that may not fit every patient’s needs.
The good news? Medicare plans have eliminated prior authorization for buprenorphine, recognizing that removing barriers leads to better patient outcomes and reduced overdose deaths.
I’m Dr. Chad Elkin, founder and Medical Director of National Addiction Specialists, board-certified in Addiction Medicine and Internal Medicine. I’ve helped hundreds of patients navigate Suboxone prior authorization requirements across Tennessee and Virginia, working to eliminate barriers that delay access to this life-saving medication.
Terms related to Suboxone prior authorization:
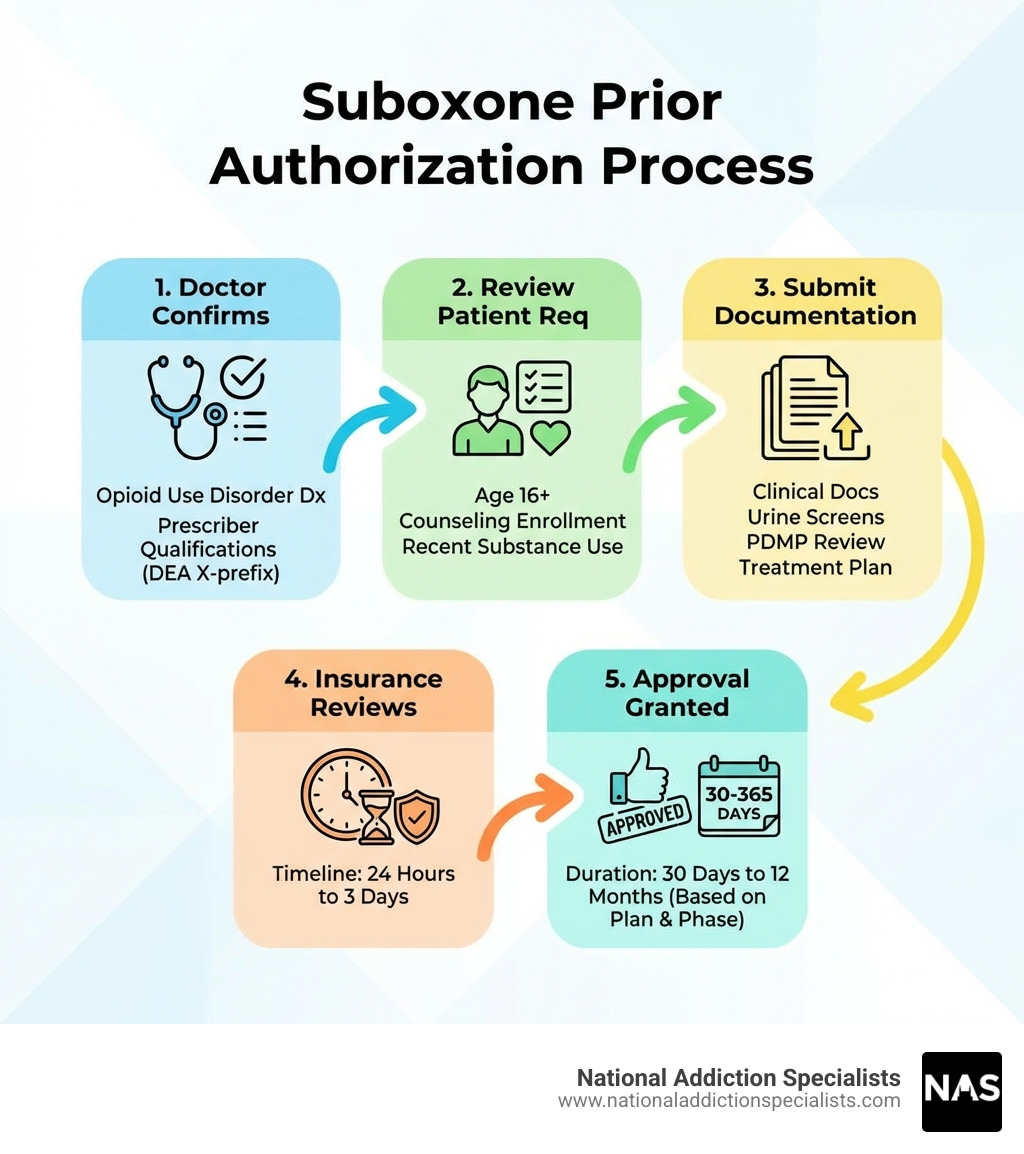
Understanding the Suboxone Prior Authorization Process
Navigating insurance can feel like trying to solve a Rubik’s Cube in the dark. When we talk about Suboxone prior authorization, we are referring to a “gatekeeper” mechanism used by insurance companies to manage how medications are used. Essentially, your insurer wants to make sure that the medication being prescribed is the most appropriate and cost-effective option for your specific condition.

The process begins when your provider submits a request to your insurance company. This request includes clinical notes documenting your diagnosis of Opioid Use Disorder (OUD) and explaining why Suboxone is necessary for your recovery. Understanding how Suboxone opioid treatment works is the first step in realizing why these clinical notes are so vital—they prove to the insurer that you are a candidate for life-saving Medication-Assisted Treatment (MAT).
Insurance companies use “utilization management” to control costs. This often means they have a list of “preferred” medications. If your doctor prescribes something not on that list, or a brand-name version when a generic is available, the Suboxone prior authorization process is triggered. In Tennessee and Virginia, Medicaid programs (like TennCare) have very specific requirements that must be met before they will pay for the medication.
Interestingly, scientific research on the impact of removing prior authorization suggests that when these barriers are taken away, patients stay in treatment longer and have better overall outcomes. Despite this, many plans still require detailed documentation regarding your insurance and pricing before they give the green light.
Key Criteria for Initial Approval
To get that initial “Yes” from your insurance provider, several boxes must be checked. First and foremost are the prescriber qualifications. Historically, under the Drug Addiction Treatment Act of 2000 (DATA 2000), physicians needed a special DEA number with an “X-prefix” to prescribe buprenorphine for addiction. While federal regulations have evolved to expand access, many insurance companies still look for these specific credentials or board certification in addiction medicine to approve the request.
Patient age is another factor; most plans cover Suboxone for patients aged 16 and older, though some follow American Academy of Pediatrics (AAP) guidelines that support use in younger adolescents when medically necessary.
When we provide Medication-Assisted Treatment, we ensure all the following criteria are documented for your insurer:
- Diagnosis of OUD: A formal diagnosis of moderate to severe opioid use disorder.
- Counseling Enrollment: Documentation that the patient is enrolled in or has been referred to behavioral health counseling.
- Urine Drug Screens (UDS): A baseline screen to identify current substance use and ensure the patient is ready for induction.
- PDMP Review: A check of the Prescription Drug Monitoring Program to ensure no conflicting opioid prescriptions are active.
Initial vs. Maintenance Authorization Requirements
| Requirement | Initial Authorization (Induction) | Maintenance Authorization (Stabilization) |
|---|---|---|
| Duration | Typically 30 days to 3 months | 6 to 12 months |
| Focus | Diagnosis and safe start | Compliance and stability |
| Drug Testing | Baseline / Weekly | Monthly or random |
| Counseling | Enrollment / Referral | Proof of ongoing participation |
| Opioid Use | Must be discontinuing active use | Abstinence from illicit opioids |
Dosage Limits and Overrides in Suboxone Prior Authorization
One of the most common hurdles in the Suboxone prior authorization process involves dosage limits. Most insurance plans have a “ceiling” for what they consider a standard daily dose.
For many plans, the standard limit is 16mg of buprenorphine per day. If your clinical needs require a higher dose—such as 24mg per day—your doctor must provide a specific clinical rationale. This might include high tolerance levels or persistent cravings at lower doses.
In some cases, such as during the initial induction or stabilization phase, a pharmacist may be able to provide a temporary override for doses up to 32mg, but this usually requires immediate follow-up documentation from the prescriber. TennCare, for example, often covers up to 16mg per day for the first six months (the induction phase) but may require a reduction to 8mg during long-term maintenance unless the doctor attests that a lower dose would risk relapse or hospitalization.
When is Buprenorphine Monotherapy Approved?
You may have heard of Subutex, which is buprenorphine without the naloxone. While Suboxone is the preferred “combination” product because the naloxone helps prevent misuse, there are specific times when buprenorphine monotherapy is medically necessary.
Comparing Suboxone vs Subutex is common among patients, but insurance companies are strict. Approval for monotherapy usually requires one of the following:
- Pregnancy: Suboxone use during pregnancy is common, but some providers and insurers prefer monotherapy to avoid fetal exposure to naloxone, though guidelines are shifting.
- Breastfeeding: Similar to pregnancy, monotherapy is often approved for nursing mothers.
- Naloxone Allergy: A documented, serious allergy (like hives or anaphylaxis) to naloxone.
- Severe Intolerance: Documented severe nausea or headaches that persisted even after trying multiple versions of the combination film and tablet.
Navigating Step Therapy and Documentation Requirements
“Step therapy” is an insurance term that basically means “try the cheapest version first.” For Suboxone prior authorization, this often means you must try and “fail” on generic buprenorphine/naloxone tablets or films before the insurance company will pay for a brand-name product like Suboxone Film or Zubsolv.
Typically, a 30-day trial of the generic is required. If you experience an adverse reaction or the generic doesn’t work effectively, your doctor can then submit clinical notes to justify the brand-name version.
At National Addiction Specialists, we utilize telemedicine in Suboxone treatment to make this documentation process seamless. We maintain digital records of your treatment plan, PDMP reviews, and progress notes, which are essential for a fast approval. According to a thematic analysis of state Medicaid requirements, having clear, organized documentation is the number one way to overcome the “restrictive surveillance” often found in state-funded plans.
Managing Challenges with Suboxone Prior Authorization
The reality is that Suboxone prior authorization can be a burden. Critics, including the American Medical Association (AMA), argue that these requirements act as access barriers that lead to treatment delays. When a patient is ready to start recovery, even a 24-hour delay can be dangerous.
The challenges often include:
- Restrictive Surveillance: Requirements for frequent, observed drug screens that can be difficult for patients with jobs or transportation issues.
- Treatment Delays: The “waiting game” at the pharmacy counter while the PA is processed.
- Increased Overdose Risk: If a patient cannot get their prescription filled, they may turn to illicit sources to avoid withdrawal.
Despite these hurdles, the benefits of Suboxone treatment for opioid addiction are undeniable. It reduces mortality, helps people regain their lives, and stabilizes brain chemistry. We work hard to manage these challenges on your behalf so you can focus on your recovery.
Make an Appointment to Treat Addiction Please don’t hesitate. Make an appointment today. https://www.nationaladdictionspecialists.com/new-patient-packet/
Authorization Periods and Reauthorization
Once your Suboxone prior authorization is approved, it isn’t “set it and forget it.” Approvals are granted for specific periods.
- Initial Period: Usually 30 days to 3 months. This covers the “induction” and “stabilization” phases where your dose might change frequently.
- Maintenance Period: Once you are stable, many plans (like UnitedHealthcare) will grant a 12-month authorization.
- Reauthorization: To keep your coverage active, your doctor must submit a reauthorization request.
During reauthorization, the insurance company looks for a “positive clinical response.” This means you are showing up for appointments, participating in counseling, and your drug screens show the presence of buprenorphine and the absence of illicit opioids. If you have questions about this, our Suboxone treatment FAQ covers many common concerns about long-term maintenance and taper strategies.
Frequently Asked Questions about Suboxone Prior Authorization
How long does it take to get a Suboxone prior authorization approved?
In most cases, insurance companies are required to respond within 24 to 72 hours. Some Medicaid plans, like those in Tennessee, aim for a 24-hour turnaround once the provider faxes the correct forms. At National Addiction Specialists, we submit these requests electronically to speed up the process as much as possible.
Can Suboxone be approved for pain management through this process?
Generally, no. Most insurance policies for Suboxone specifically state that it is only covered for the treatment of Opioid Use Disorder. If a doctor prescribes buprenorphine solely for chronic pain, it usually falls under a different set of “long-acting opioid” prior authorization rules, which are often much stricter and require failure of non-opioid treatments first.
What happens if I test positive for other substances during treatment?
A positive drug screen for substances like benzodiazepines or other opioids does not necessarily mean your Suboxone prior authorization will be revoked, but it will require your doctor to document a plan of action. Most guidelines suggest that treatment should not be discontinued due to a relapse, but rather the treatment plan should be adjusted (e.g., more frequent counseling or a dose adjustment).
Conclusion
Getting your Suboxone prior authorization approved doesn’t have to be a nightmare. By working with an expert team that understands the specific requirements in Tennessee and Virginia, you can bypass the red tape and get the medication you need to stay safe and healthy.
At National Addiction Specialists, we provide telemedicine-based care that fits your life. Whether you are in Brentwood, Virginia Beach, or anywhere in between, our expert providers are here to help. We accept Medicaid and Medicare and specialize in creating personalized recovery plans from the comfort of your home. Check out our insurance and pricing page to see how we can help you start your journey today.
Make an Appointment to Treat Addiction Please don’t hesitate. Make an appointment today. https://www.nationaladdictionspecialists.com/new-patient-packet/
This article was medically reviewed by: Chad Elkin, MD, DFASAM is a board-certified addiction medicine physician, founder, and Chief Medical Officer of National Addiction Specialists, dedicated to treating substance use disorders. A Distinguished Fellow of the American Society of Addiction Medicine (ASAM), Dr Elkin currently serves as President of the Tennessee Society of Addiction Medicine (TNSAM) and has held various leadership roles within the organization. Dr Elkin chairs ASAM’s Health Technology Subcommittee and is an active member of its Practice Management and Regulatory Affairs Committee, State Advocacy and Legislative Affairs Committee, and other committees. He also serves on the planning committee for the Vanderbilt Mid-South Addiction Conference. Committed to advancing evidence-based policy, Dr Elkin is Chairman of the Tennessee Association of Alcohol, Drug, & Other Addiction Services (TAADAS) Addiction Medicine Council, which collaborates with the TN Department of Mental Health & Substance Abuse Services (TDMHSAS). He has contributed to numerous local, state, and national task forces, helping develop professional guidelines, policies, and laws that align with best practices in addiction medicine. His work focuses on reducing addiction-related harm, combating stigma, and ensuring access to effective treatment. Passionate about the field of addiction medicine, he remains dedicated to shaping policy and enhancing patient care.
Suboxone® and Subutex® are a registered trademark of Indivior UK Limited. Any mention and reference of Suboxone® and Subutex® in this website is for informational purposes only and is not an endorsement or sponsorship by Indivior UK Limited.